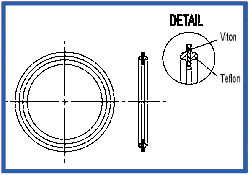
Orifice Plate – Viton Stainless Steel (K186)
THE FINER DETAILS...
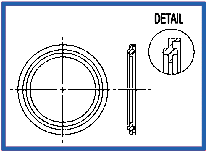
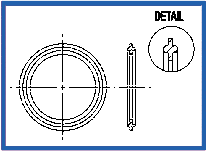
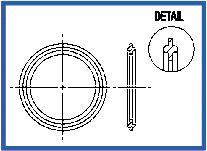
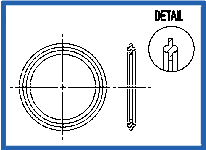
Orifice Plate – Viton Stainless Steel (K186)
Biobore Elastomer & Thermoplastic Characteristics
All materials are approved by: 3A, USDA, and comply with FDA’s Code of Federal Register – Title 21, Part 177.
PTFE GASKETS – LIMITED USE NOTE
PSI Service Ratings for clamps DO NOT APPLY to 1” and above connections incorporating PTFE gaskets. PTFE is virtually incompressible and tends to “cold flow” under normal operating conditions. PTFE gaskets, when used with any standard clamp, are likely to leak at pressures below the published service ratings.
To avoid possible leakage problems, we recommend consulting SFL before considering the use of PTFE material. This limitation does not apply to 0.5” and 0.75” Tube OD fittings and connections, due to the recessed, full-containment design of Fractional end connections.
ENVELOPE GASKETS
Consists of a PTFE outer jacket sandwiching a viton insert thus imparting some compressibility together with the excellent heat and steam resistant properties.
Download SFL Catalogue
We are proud to be a family-owned and operated business and the only UK manufacturer of BPE compliant fittings. SFL also stocks and distributes the full range of accredited ASME BPE tube and fittings, in addition to providing a full bespoke fabrication service.
Certification
Stainless Fittings (SFL) and Dairy Pipe Lines (DPL) are manufacturers of a wide range of stainless steel fittings, valves and bespoke fabricated units for the Bio-Processing, Pharmaceutical, Food, and Drink industries. The companies are regularly assessed to the ISO 9001:2015 Quality Management standard.
As part of our ongoing commitment to quality, the company has invested in systems that enable the traceability of materials used in both standard and fabricated products. These systems provide for the recording and reporting of quality and inspection data from various points on the manufacturing cycle.
Products meeting the ASME Bio Processing Equipment standard (BPE), will always be accompanied by full material certificates either with the goods or by email, or both if required. Where needed, SFL will provide a full quality dossier to cover project work.
The following process certificates can be made readily available when specified with the enquiry:
- Welding Certificates
- Welder approvals and weld procedures for each weld type and size of material.
- A weld plan and weld log for each item including any weld filler rod if used.
- CD Video files of each internal weld by its ID and part number immediately after welding.
- Welding machine calibration certificates.
- Orbital weld machine setting parameters and / or pre-weld coupons.
Surface Finish Certificates
A surface finish print-out for each item. A cleaning procedure document. A passivation certificate or electro polish certificate as required.
Functional Test Certificates
We will supply a pressure test certificate and pressure gauge calibration certificate to help with your obligations under the Pressure Equipment Directive. Additionally a dye penetrant test certificate or radiographic image and report can be supplied.
Declaration Documentation for EC Directives
Our valves meet the Pressure Equipment Directive (PED) and in some cases the Machinery Safety Directive (for powered valves). Selected valves are manufactured to comply with the ATmospheric EXplosive directive (ATEX). A safety declaration will be supplied in accordance with these directives.
Additionally, DPL offer a Certificate of Conformance for all products supplied. Certificates depicting seal type, FDA schedule number and operational pressure test are also available for all valves.
Quality
Stainless Fittings (SFL) and Dairy Pipe Lines (DPL) are manufacturers of a range of stainless steel fittings, valves and bespoke fabricated units for the bio-processing, pharmaceutical and food industries. The companies are regularly assessed to the ISO 9001:2015 Quality Management standard.
The products provided by the company meet the stringent statutory and regulatory requirements demanded by the marketplace. Inspections and product tests are carried out at regular stages of manufacture allowing comprehensive quality records to be maintained. These records can then be used for analysis as part of our commitment to ongoing process and product improvements. As part of this process, bi-monthly reviews are in place to ensure the quality system continues to support our business objectives.
With the processes of the Quality Management System defined and implemented, one major role of the management team is to adapt these processes to meet changing requirements in the industry and to enhance the business wherever this results in a benefit to our customers.
Our initial contact with our customers is normally through the Sales team. Here your precise requirements are assessed and stock availability discussed with you in order to meet your needs. Where you require us to make use of our fabrication facility, we take the time to understand your exact requirements even further.
After being sure that your sales contact has a clear understanding of your requirements, we conduct a Contract Review Meeting to ensure that the entire team responsible for manufacturing your order are all clear on what is needed.
We will contact you where we need further information or where we need specific points being clarified. All agreements are documented so you can be sure we have your requirements on board.
Material traceability is of prime importance to us, and therefore our internal bespoke systems and material identification allows us to trace material back to its origin.
SFL and DPL offer a wide range of certification options to include Certificates of Conformance, Original Material Certificates, EP or Passivation Certs, Weld maps, Qualifications and Procedures to name a few. Just discuss your requirements with us – our Mission is your Peace of Mind.
Click one of these links to view or download our EU Directive Statements for: